|
For example, in the reaction of hydrogen (H₂) with oxygen (O₂) to form water (H₂O), the chemical equation is: It shows the reactants (substances that start a reaction) and products (substances formed by the reaction). 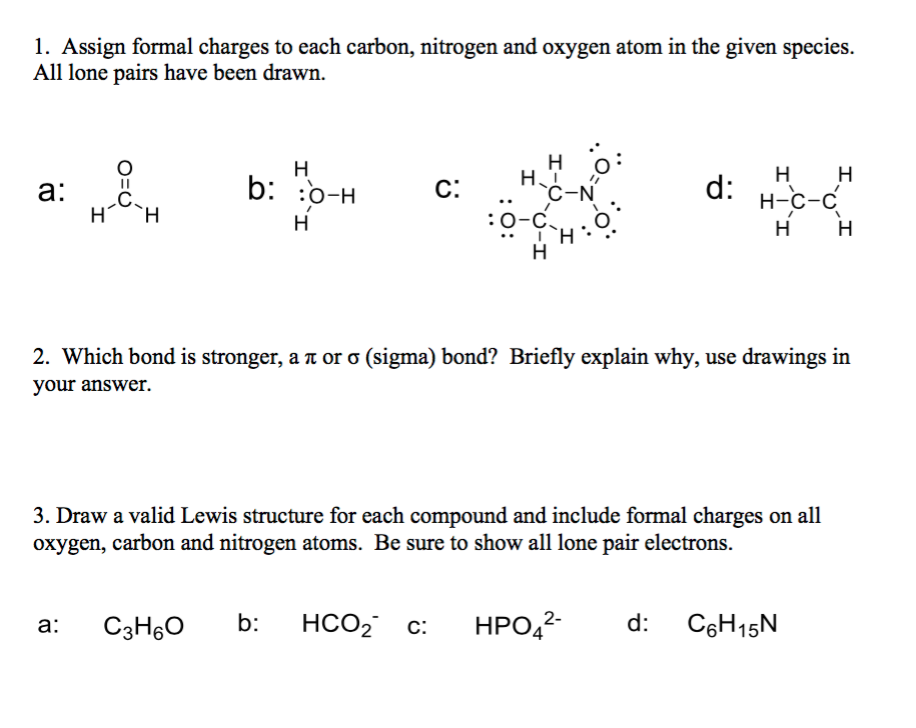
calcium hydroxide + carbon dioxide = calcium carbonate + waterĮxamples of the chemical equations reagents (a complete equation will be suggested):Ī chemical equation represents a chemical reaction.PhCH 3 + KMnO 4 + H 2SO 4 = PhCOOH + K 2SO 4 + MnSO 4 + H 2O.To enter an electron into a chemical equation use + H 2O.Compare: Co - cobalt and CO - carbon monoxide Always use the upper case for the first character in the element name and the lower case for the second character.Ğxamples: Fe, Au, Co, Br, C, O, N, F.Enter an equation of a chemical reaction and click 'Balance'.Instructions on balancing chemical equations: Please tell about this free chemistry software to your friends! In such cases, chemists usually define a standard by arbitrarily assigning a numerical value to one of the quantities, which allows them to calculate numerical values for the rest.įigure 1.6.2 Determining Relative Atomic Masses Using a Mass Spectrometer. We will encounter many other examples later in this text. It is actually rather common in chemistry to encounter a quantity whose magnitude can be measured only relative to some other quantity, rather than absolutely. Thus it is not possible to calculate absolute atomic masses accurately by simply adding together the masses of the electrons, the protons, and the neutrons, and absolute atomic masses cannot be measured, but relative masses can be measured very accurately. By measuring the relative deflection of ions that have the same charge, scientists can determine their relative masses (Figure 1.6.2). The extent of the deflection depends on the mass-to-charge ratio of the ion. When an electric field is applied, the ions are accelerated into a separate chamber where they are deflected from their initial trajectory by a magnetic field, like the electrons in Thomson’s experiment. 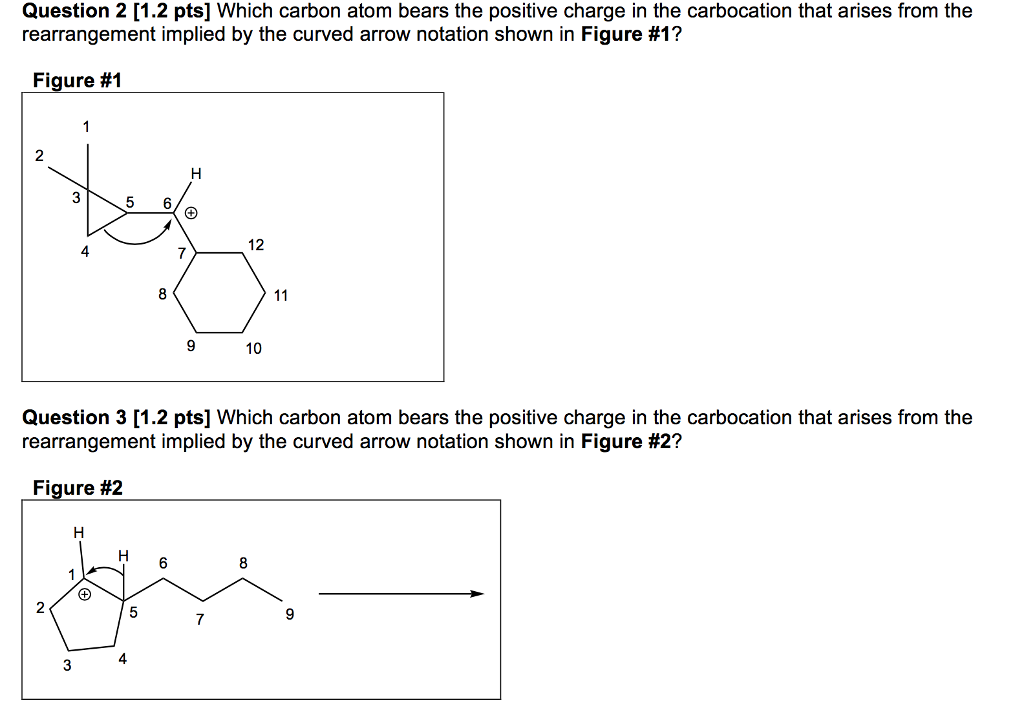 
First, electrons are removed from or added to atoms or molecules, thus producing charged particles called ions. The technique is conceptually similar to the one Thomson used to determine the mass-to-charge ratio of the electron. Scientists can measure relative atomic masses very accurately, however, using an instrument called a mass spectrometer. We can easily calculate the binding energy from the mass difference using Einstein's formula E=mc 2.īecause atoms are much too small to measure individually and do not have a charge, there is no convenient way to accurately measure absolute atomic masses. Although the difference in mass is small, it is extremely important because it is the binding energy of the nucleus. For example, the ratio of the masses of 1H (hydrogen) and 2H (deuterium) is actually 0.500384, rather than 0.49979 as predicted from the numbers of neutrons and protons present. Br\) or, more commonly, 79Br and 81Br.Īlthough the masses of the electron, the proton, and the neutron are known to a high degree of precision (Table 1.5.1), the mass of any given atom is not simply the sum of the masses of its electrons, protons, and neutrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
